The semimetals lie along the diagonal line dividing metals and nonmetals. Conversely, nonmetals have high electronegativities, and they therefore tend to gain electrons in chemical reactions to form compounds in which they have negative oxidation states. Because metals have relatively low electronegativities, they tend to lose electrons in chemical reactions to elements that have relatively high electronegativities, forming compounds in which they have positive oxidation states. The diagonal line in Figure 21.1 "Summary of Periodic Trends in Atomic Properties" separates the metals (to the left of the line) from the nonmetals (to the right of the line). 
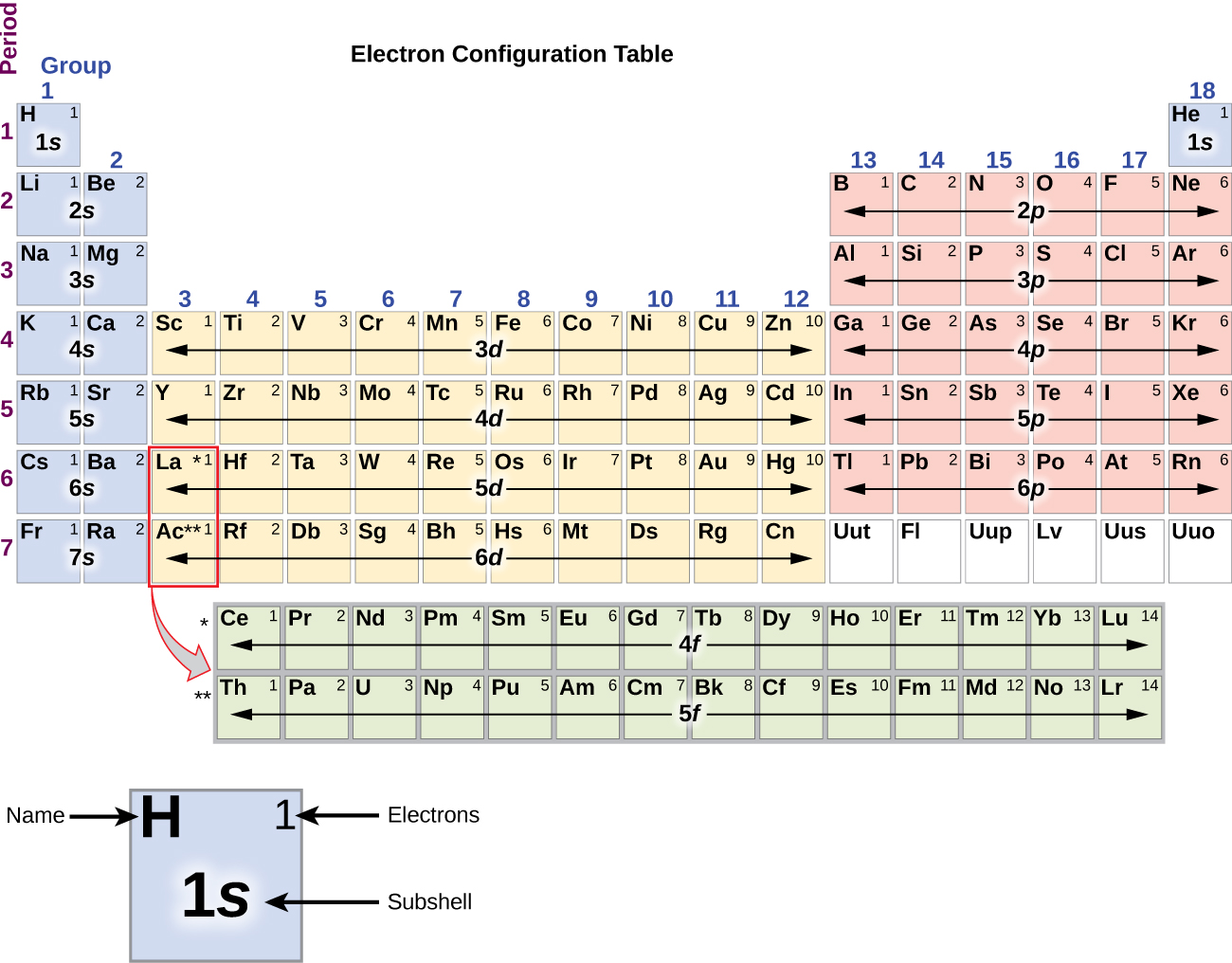
( Z eff), which increases from left to right and from top to bottom in the periodic table ( Figure 6.29 "Orbital Energy Level Diagram for a Typical Multielectron Atom"). Recall from Chapter 7 "The Periodic Table and Periodic Trends" that these trends are based on periodic variations in a single fundamental property, the effective nuclear charge The nuclear charge an electron actually experiences because of shielding from other electrons closer to the nucleus. The most important periodic trends in atomic properties are summarized in Figure 21.1 "Summary of Periodic Trends in Atomic Properties". The same factors also lead to periodicity in valence electron configurations, which for each group results in similarities in oxidation states and the formation of compounds with common stoichiometries. 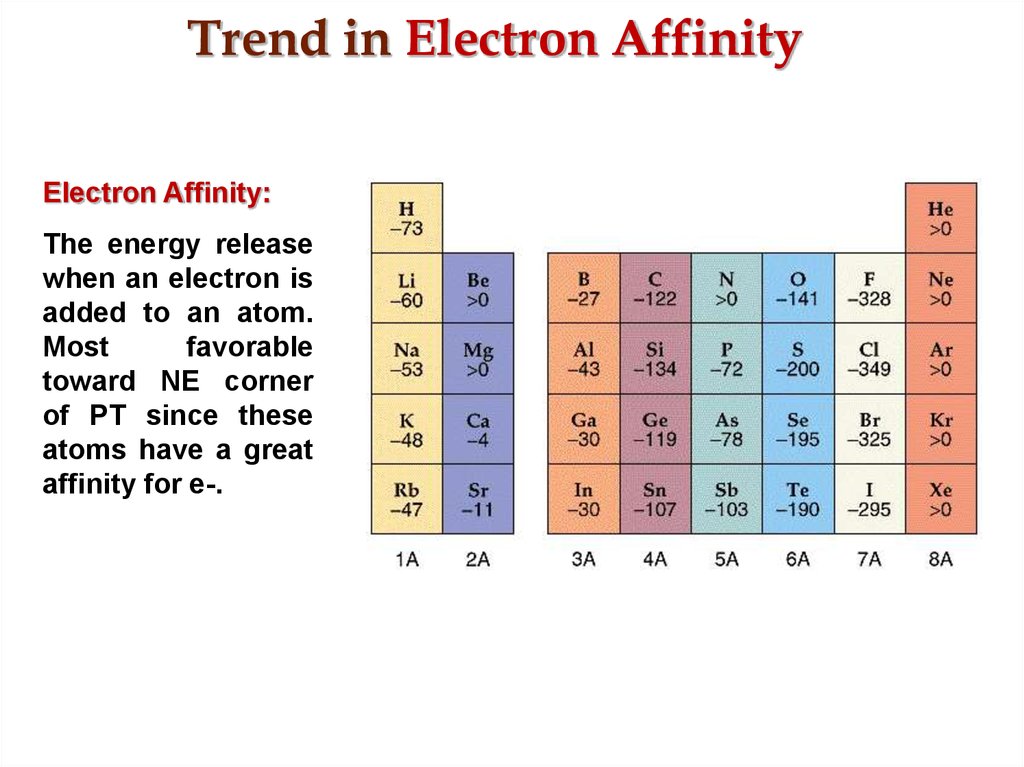
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
